Question: 100% NCERT covered from Particular Keyword.Explanations are 100% from NCERT | [Difficult level: Easy] From NCERT NEET 2012.
In Freundlich adsorption isotherm, the value of 1/n is -
1. Between 0 and 1 in all cases.
2. Between 2 and 4 in all cases.
3. 1 in case of physical adsorption.
4. 1 in case of chemisorption.
Answer:1. Between 0 and 1 in all cases.
Question: 100% NCERT covered from Particular Keyword.Explanations are 100% from NCERT | [Difficult level: Easy] From NCERT NEET Derived Question.
Answer:b
Question: 100% NCERT covered from Particular Keyword.Explanations are 100% from NCERT | [Difficult level: Easy] From NCERT NEET Derived Question.
According to Freundlich adsorption isotherm, the amount
of gas adsorbed at very high pressure
(a) reaches a constant limiting value
(b) goes on increasing with pressure
(c) goes on decreasing with pressure
(d) increase first and decreases later with pressure
Answer:(a) reaches a constant limiting value
The process of adsorption taking place at a constant temperature but the pressure varies is called adsorption isotherm. It is of two types –
(a) Freundlich’s adsorption isotherm
(b) Langmuir adsorption isotherm.
Langmuir adsorption isotherm:
The Langmuir adsorption isotherm is used to describe the equilibrium between adsorbate and adsorbent system, where the adsorbate adsorption is limited to one molecular layer at or before a relative pressure of unity is reached.
Langmuir’s adsorption isotherm is based on the kinetic theory of gases. Langmuir considered adsorption to consist of the following two opposing processes.
(i) Adsorption of the gas molecules on the surface of the solid.
(ii) Desorption of the adsorbed molecules from the surface of the solid.
In the Langmuir model, the adsorbent surface is considered to possess a number of active interaction sites for adsorption. Langmuir derived a relation between adsorbed material and its equilibrium concentration, i.e.
Where x = mass of solute adsorbed
m = Mass of adsorbent
Equilibrium concentration of solute
Equilibrium constant
Thus, the adsorption can be given by the following Langmuir expression,
Where x = mass of gas in gm
m = Mass of adsorbent in gm
p = Pressure
a and b = Langmuir parameter
Validity of Langmuir adsorption isotherm:
(a) At moderate pressure
(b) At low pressure; bp < < 1
(c) At high pressure
constant
The Langmuir expression can be represented as under
On taking reciprocal,
Since, a and b are constant. Hence, graph between and p should be a straight line and the slope is equal to , intercept is equal to .
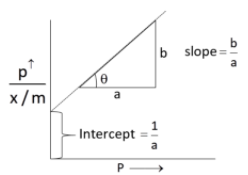
According to Langmuir adsorption, the amount of gas adsorbed at a very high pressure, reaches a constant limiting value.
Question: 100% NCERT covered from Particular Keyword.Explanations are 100% from NCERT | [Difficult level: Easy] From NCERT NEET Derived Question.
Which of the following curves is in accordance with
Freundlich adsorption isotherm ?
Answer: c
Question: 100% NCERT covered from Particular Keyword.Explanations are 100% from NCERT | [Difficult level: Easy] From NCERT NEET Derived Question.
(a) Assertion is correct, reason is correct; reason is a correct
explanation for assertion.
(b) Assertion is correct, reason is correct; reason is not a
correct explanation for assertion
(c) Assertion is correct, reason is incorrect
(d) Assertion is incorrect, reason is correct.
Answer:(c) Assertion is true, reason is false. When several lines have the same value of 1/ n , then the lines by which their adsorption isotherms can be represented will be parallel and will not meet at a point.
Question: 100% NCERT covered from Particular Keyword.Explanations are 100% from NCERT | [Difficult level: Easy] From NCERT NEET Derived Question.
In the adsorption of a gas on solid, Freundlich isotherm is
obeyed. The slope of the plot is zero. Then the extent of
adsorption is
(a) directly proportional to the pressure of the gas
(b) inversely proportional to the pressure of the gas
(c) directly proportional to the square root of the pressure
of the gas
(d) independent of the pressure of the gas
Answer:d
What is an adsorption isotherm? Describe Freundlich adsorption isotherm.
The variation in the amount of gas adsorbed by the adsorbent with
pressure at constant temperature can be expressed by means of a
curve termed as adsorption isotherm.
Freundlich adsorption isotherm: Freundlich, in 1909, gave an
empirical relationship between the quantity of gas adsorbed by unit
mass of solid adsorbent and pressure at a particular temperature. The
relationship can be expressed by the following equation:
The validity of Freundlich isotherm can be
verified by plotting log
x/m
on y-axis (ordinate)
and log p on x-axis (abscissa). If it comes to be
a straight line, the Freundlich isotherm is valid,
otherwise not.
Freundlich isotherm explains the behaviour
of adsorption in an approximate manner. The
factor 1/n can have values between 0 and 1
(probable range 0.1 to 0.5).






